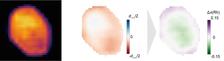
The platinum-rhodium nanoparticle studied under the X-ray beam and scanning electron microscope. The particle is about 143 nanometres wide and 213 nanometres long. (Image: DESY, Arno Jeromin.)
A team of international scientists, also from DESY, has studied individual nanoparticles made from an alloy of platinum and rhodium, two catalytic materials, under conditions encountered in technical applications. The analysis using X-rays and a scanning electron microscope shows how these two metals respond to different external conditions and accumulate on the surface of the nanoparticle in the process. The team headed by Hoydoo You from the Argonne National Laboratory in the USA is presenting its study in the journal Physical Review Letters.
“The results can help us to understand how to keep the catalytic nanoparticles active for longer,” explains co-author Thomas Keller, from DESY NanoLab. Catalysts are substances which allow a chemical reaction to occur but which are not consumed in the process. They have a wide range of applications in chemistry, including the catalytic converters installed in car exhaust systems, where they promote the oxidation of harmful substances such as carbon monoxide and nitrous oxides into less critical compounds. Both rhodium and platinum are of substantial technological significance and frequently used in catalysts, also in mixtures.
For their study, the scientists at DESY NanoLab came up with a way of manufacturing alloy nanoparticles of a size suitable for making X-ray measurements. To do so, platinum was first deposited on a substrate in a vacuum, using heat treatment to adjust their size, after which the platinum was coated with rhodium at high temperatures, in order to achieve the optimum level of mixing. Afterwards a nanoparticle was selected in the scanning electron microscope and individual markers were applied. In the X-ray experiment, the sample was then exposed to a range of different temperatures and atmospheres of varying gaseous compositions, while the previously marked particle was studied in the X-ray beam.
The scientists used X-rays from the Advanced Photon Source in Argonne to determine the spatial distribution of the displacements in the crystal lattice, making use of the fact that the atoms of the two elements differ slightly in size. This means they can switch positions within the crystal lattice and thereby migrate through the particle – driven by external factors such as the temperature or gas composition. The local crystal lattice therefore shifts slightly, and the measured displacement can be translated into the local change in the concentration of the rhodium atoms.
“In an oxygen-rich atmosphere, the rhodium atoms migrate through the crystal lattice to the surface of the particle because they can be oxidised more easily than platinum,” explains Henning Runge, a PhD student at DESY and the University of Hamburg who worked on the experiment in Argonne. “This had already been observed in crystals made from platinum-rhodium alloys, but it was not clear how the two metals behaved in nanoparticles, where additional factors such as the surface energy become more important.” Since catalytic reactions occur on the surface of the metal, one tries to shape the catalyst into the smallest possible particles or structures, so as to increase the surface area and with it the efficiency of the catalyst. The particle studied was about 140 by 210 nanometres (millionths of a millimetre) across.
Being a very active catalyst, rhodium is of considerable interest for many reactions. However when it becomes fully oxidised on the surface of the catalyst, it becomes inactive and is no longer available for catalysis. This is why catalysts should ideally be tailored to fit the specific application, depending on the prevailing conditions in the environment.
“Under reductive conditions, for example in a hydrogen-rich atmosphere, the rhodium atoms return from the oxidised state back into the crystal lattice,” as the principal author Tomoya Kawaguchi from the Argonne National Laboratory and Tohoku University in Japan explains.
“We are now trying to understand how such a nanoparticle can best be kept active,” says Keller. “The method we have developed for marking them allows us for the first time to characterise individual nanoparticles and to proceed to analyse them further at DESY NanoLab, instead of studying a large number of particles and looking at the mean, as is normally the case. This means that systematic effects can be studied directly.”
The experiments were conducted by the Argonne National Laboratory, Tohoku University, the University of Hamburg, the National Research Nuclear University MEPhI in Moscow and by DESY.
(from DESY News)
Reference:
Tomoya Kawaguchi, Thomas F. Keller, Henning Runge, Luca Gelisio, Christoph Seitz, Young Y. Kim, Evan R. Maxey, Wonsuk Cha, Andrew Ulvestad, Stephan O. Hruszkewycz, Ross Harder, Ivan A. Vartanyants, Andreas Stierle, and Hoydoo You; Gas-induced segregation in Pt-Rh alloy nanoparticles observed by in situ Bragg coherent diffraction imaging; Physical Review Letters, (2019); DOI: 10.1103/PhysRevLett.123.246001