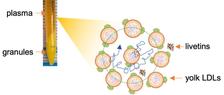
Left: After centrifugation, egg yolk separates into a translucent yolk plasma fraction and an opaque granule fraction. Right: Schematic of LDL nanoparticles (fat carriers) in yolk plasma. Surrounded by neighbouring particles and livetins (yolk-plasma proteins), LDLs can become temporarily trapped in nanoscale “cages”. The blue line illustrates the trajectory of the central LDL molecule in a cage (dotted circle) formed by other LDLs and livetins. Longer-range motion resumes only when the local neighbourhood reorganises—a process known as cage relaxation. (Illustration: University of Siegen)
Egg yolk may appear runny and uniform but on the nanoscale it is one of the most crowded biological fluids in nature. Packed with proteins and fats, it serves as a dense storage reservoir for a developing embryo. Yet, the tiny particles responsible for transporting these nutrients—low-density lipoproteins (LDLs)—must remain mobile enough to reach their destinations. How they navigate this complex "nanoscale traffic jam" has long puzzled scientists.
In a study published in the Proceedings of the National Academy of Sciences (PNAS), an international team led by Christian Gutt and Nimmi Das Anthuparambil from the University of Siegen and DESY has uncovered the physical principles governing lipoprotein motion within egg yolk plasma, the translucent, fluid component of egg yolk. Using an advanced X-ray technique at the European XFEL in Schenefeld, the scientists observed fat-transporting particles on microsecond timescales—taking snapshots over a million times faster than the blink of an eye.
Watching motion that was previously invisible
Due to the extreme crowding inside yolk, conventional methods have been unable to capture the movement of these particles. To overcome this, the researchers employed megahertz X-ray photon correlation spectroscopy (MHz-XPCS) at the European XFEL. "The unique pulse structure of the European XFEL lets us probe structural dynamics on microsecond timescales," explains Anders Madsen, leading scientist and responsible for the Materials Imaging and Dynamics (MID) instrument at European XFEL. "MHz-XPCS captures these rapid, subtle motions inside crowded biological fluids in real time—dynamics that otherwise remain hidden." For the first time, the team directly observed how these fat particles move, collide and interact with their neighbours in real time. The LDLs navigate a tightly packed suspension of other LDLs and soluble proteins called livetins. These are forming temporary ”cages” around the LDLs that prevent free diffusion. The LDLs become trapped, rattling within these confines before slowly escaping. “Data analysis in situ during the experiment is required to cope with the enormous amount of data generated by MHz-XPCS, and together with our colleagues we cracked the nut to enable this live information” adds Johannes Möller, instrument scientist at MID.
Unexpected slowness
This caging dramatically slows LDL movement—by up to 100 times compared to dilute solutions. Surprisingly, despite this slowdown, egg yolk behaves not as a solid but as a thick yet flowing liquid. "This sluggish liquid state seems to be exactly what biology needs," says lead author Nimmi Das Anthuparambil. "It keeps nutrients safely stored while still allowing controlled transport when the embryo requires them." One surprising finding is that LDL diffusion in dense yolk does not follow conventional behaviour. Normally, diffusion slows predictably with increasing fluid viscosity, described by the Stokes–Einstein relation. However, in this crowded environment, soft LDLs move far more slowly than expected, indicating a breakdown of this classical law. The observed caging cannot fully explain the slowness. "This means new models are needed to understand transport in complex biological fluids," explains principal investigator Christian Gutt from University of Siegen. The researchers found that the slowdown cannot be explained by viscosity alone; instead, particle softness and long-range hydrodynamic interactions play a critical role, causing classical hard-sphere diffusion theories to fail in these crowded conditions.
Why this matters beyond egg yolk
Understanding how particles move in dense biological fluids has implications far beyond embryonic development. Similar crowded environments exist inside living cells, protein-rich solutions and in drug-delivery systems that use lipoproteins as natural nanocarriers.
By combining ultrafast X-ray experiments with a new theoretical framework, this study establishes a quantitative link between microscopic structure and large-scale transport in crowded soft-matter systems. This approach can be applied to diverse biological and technological materials—ranging from cell interiors to pharmaceutical formulations. Low-density lipoproteins are promising natural drug carriers, and their ability to move efficiently in crowded spaces directly influences their efficiency. “Life depends on movement in crowded environments—molecules and particles navigating constraints without the system turning solid,” says Sakura Pascarelli, Scientific Director at European XFEL. “Studies like this show how advanced X-ray methods can reveal the physics that makes life possible.”
This study was conducted as part of a long-term project at the MID instrument. The work involved close collaboration between the Universities of Siegen, Tübingen, Hamburg, Stockholm, the Technical University of Dortmund, ESRF—The European Synchrotron in Grenoble and DESY. The data management and analysis workflow was developed in the context of the DAPHNE4NFDI initiative, ensuring FAIR-compliant handling of experimental data. This research was supported by the Swedish-German Röntgen-Angström Cluster (RAC), funded by the Bundesministerium für Forschung, Technologie und Raumfahrt (BMFTR).
(from DESY/EuXFEL News)
Reference
Nimmi Das Anthuparambil, Michelle Dargasz, Sonja Timmermann, Christian Gutt. "Lipoprotein diffusion in dense yolk plasma is governed by softness, hydrodynamics, and caging: Insights from MHz-XPCS."
Proceedings of the National Academy of Sciences PNAS (2025) DOI:
10.1073/pnas.2519681123